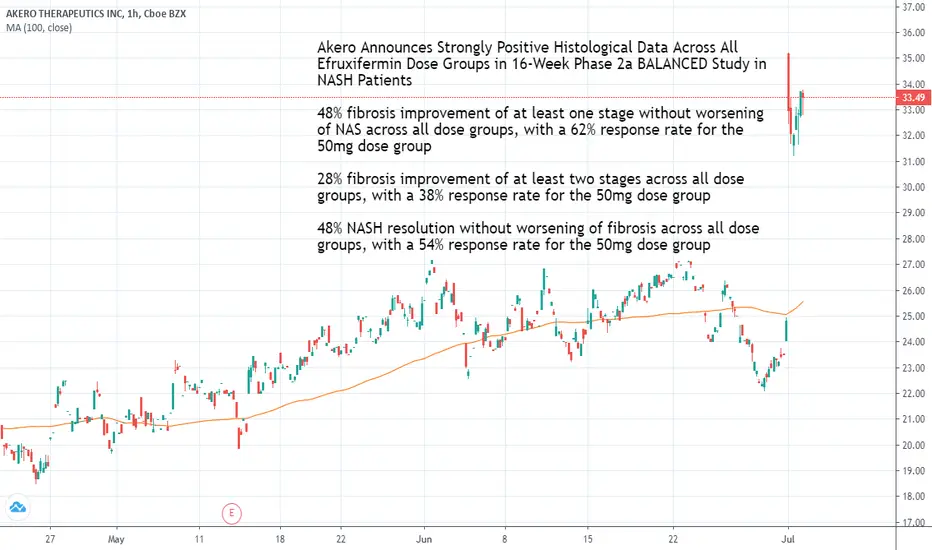
Akero Announces Strongly Positive Histological Data Across All Efruxifermin Dose Groups in 16-Week Phase 2a BALANCED Study in NASH Patients
48% fibrosis improvement of at least one stage without worsening of NAS across all dose groups, with a 62% response rate for the 50mg dose group
28% fibrosis improvement of at least two stages across all dose groups, with a 38% response rate for the 50mg dose group
48% NASH resolution without worsening of fibrosis across all dose groups, with a 54% response rate for the 50mg dose group
ir.akerotx.com/news-releases/news-release-details/akero-announces-strongly-positive-histological-data-across-all
48% fibrosis improvement of at least one stage without worsening of NAS across all dose groups, with a 62% response rate for the 50mg dose group
28% fibrosis improvement of at least two stages across all dose groups, with a 38% response rate for the 50mg dose group
48% NASH resolution without worsening of fibrosis across all dose groups, with a 54% response rate for the 50mg dose group
ir.akerotx.com/news-releases/news-release-details/akero-announces-strongly-positive-histological-data-across-all
Clause de non-responsabilité
Les informations et publications ne sont pas destinées à être, et ne constituent pas, des conseils ou recommandations financiers, d'investissement, de trading ou autres fournis ou approuvés par TradingView. Pour en savoir plus, consultez les Conditions d'utilisation.
Clause de non-responsabilité
Les informations et publications ne sont pas destinées à être, et ne constituent pas, des conseils ou recommandations financiers, d'investissement, de trading ou autres fournis ou approuvés par TradingView. Pour en savoir plus, consultez les Conditions d'utilisation.